How Do You Know if Distillation Is Successful
Experiment 5
Liquids: Distillation, Boiling Points, and the Fermentation and Distillation of Ethanol
Objectives
Distillations are performed to isolate volatile chemicals from not-volatile or less volatile chemicals. This experiment addresses how distillations (both uncomplicated and fractional) tin can be used to isolate chemical in pure class. In improver, humid points and other physical properties tin be adamant, including refractive index and density of purified materials. Nosotros also setup a fermentation to isolate ethanol from these mixtures.
Groundwork
Parts A and B will exist performed during the kickoff laboratory period. Part C, which is the distillation of ethanol from a fermentation mixture, will exist performed during the second laboratory period.
Techniques and procedures that you will perform during this experiment include:
- Simple distillation
- Fractional distillation
- Refractive alphabetize determination
This experiment is performed in three parts over two days. Parts A & B are performed during the first day. Part C is performed during the 2nd day of lab. The fermentation setup is done one week prior to the beginning of Role C.
Part A: Purify 2-propanol by simple distillation. This technique shows how to purify, by distillation, a volatile organic compound. This procedure is skillful for purification of volatile compounds from compounds that are not volatile, simply non very successful for mixtures of compounds, each of which is volatile. Function B describes how to fractionate these types of mixtures using fractional distillation.
Part B: Separate ethanol from water by partial distillation. Compounds which are volatile volition each distill at temperatures well below their actual humid points (e.g., remember the vapor pressure of water?). We need to accept a procedure that allows for more efficient separation of these volatile liquids, and this is the process of fractional distillation.
Role C: We will prepare a fermentation of glucose to produce ethanol. The ethanol from this fermentation will exist isolated by fractional distillation (using the process described in Part B). You must repeat your fractional distillation, in order to remove water, which, unfortunately predominates during the starting time fractional distillation. One time you have repeated your fractional distillation, yous tin can determine the amount of alcohol in your final distillate sample.
In many of the previous experiments that nosotros have performed, (eastward.g., the experiments involving extraction, separation, Re-crystallization, and chromatography), you lot accept learned that a pharmacist must exist able to exploit the specific physical backdrop of the components of a mixture in gild to efficiently separate and purify the desired chemical compound. The most common method for separating and purifying volatile liquids is distillation, which makes employ of the specific boiling points of the liquid components in the mixture.
When there is merely one volatile liquid, or when 1 of the liquids has a boiling signal well below the others, a elementary distillation tin be used. However, if there are two or more liquid components, which have humid points near each other, a fractional distillation must be used (the theory of distillations is discussed in Chapter 35 of Zubrick).
In Function A of this experiment you will carry out a simple distillation of 2-propanol (isopropyl booze), which has been contaminated with a not-volatile impurity (a dye). In Function B, you will carry out a fractional distillation of a 50:50 (v/v) mixture of ethanol and water.
During each of these experiments, you will determine the boiling signal of the volatile organic compound during distillation. You will monitor the temperature that the volatile liquid has, in the gas phase using a thermometer. This temperature should remain constant, and should reflect the bodily boiling point of the chemical under the atmospheric weather of the mean solar day. Report this boiling indicate equally part of your data.
Procedure
Role A: Uncomplicated Distillation of ii-propanol
Condom:2-propanol is a highly flammable liquid and a severe centre irritant -- no flames will exist allowed in lab while it is in apply. Equally for every experiment, goggles must be worn, fifty-fifty though you may not actually be working the chemicals, if in that location is anyone using two-propanol in the lab.
Follow the instructions in capacity 19 and twenty of Zubrick for setting up a simple distillation appliance, although the instructor will go through the setup, and the use of your organic kit. You volition use a 50-mL circular lesser flask equally the distillation pot, and a 100-mL round bottom flask as the receiver. As a standard rule, anytime you are humid an organic compound, you will always include a few (not a handful!) humid stones to keep the solution humid smoothly!
Using a beaker (not a graduated cylinder) obtain about xxx-40 mL of "impure" two-propanol (this sample of 2-propanol has had a small amount of a soluble, non-volatile dye added to information technology equally an impurity). Add the 2-propanol to the distillation pot (never pour anything through a ground-glass opening without using a funnel), add the boiling stones, and begin the distillation (remember to turn on the cooling water before you turn on the estrus). Collect your distillate in a pre-weighed graduated cyclinder (10-mL or 25-mL or 50-mL cylinder).
Once the temperature starts to rise above room temperature, you should start to record the thermometer reading every minute.
You should get-go to record the temperature at any time up to when the solution starts to boil. Tape every minute the temperature you read. Continue to do your distillation until y'all accept collected about 20 mL of distillate. Exist certain that the distillation pot never goes dry (never allow a heated flask go dry!).
Y'all must plot your information by hand using graph newspaper, or y'all can utilise Excel or another graphing program for a graph for inclusion in your lab notebook and written written report.
Mensurate the volume of the distillate collected. Using a pre-weighed graduated cylinder (10-mL or 25-mL or 50-mL cylinder), you should decide the density of the nerveless distillate. You will also determine the refractive alphabetize of your distilled liquid. Your instructor will describe how a refractive index is adamant.
Dispose of your liquid, and any liquid remaining in the distillation pot, in the liquid waster. Exist sure to brand certain that no boiling stones are deposited into the liquid waste. Put the boiling stones in the solid waste container.
Part B: Fractional Distillation of an Ethanol/Water Mixture
Safety: Ethanol is a flammable liquid and an irritant; avert contact and inhalation -- wear gloves while handling it. No flames will exist allowed in lab while ethanol is in use. Goggles must be worn whenever anyone is using chemicals.
Set a fractional distillation apparatus every bit demonstrated by your instructor. Utilise glass chaplet to pack the fractionating column (your instructor will demonstrate how to pack the column). Try adding some glass beads straight to your fractionating cavalcade. If the drinking glass chaplet stay in the cavalcade, there is no problem, only if any beads become through, endeavor calculation a larger amount of glass bead, and their packing inside the fractionating cavalcade should allow them to stay in place. Do not e'er use drinking glass wool or anything likewise chaplet in the fractionating columns.
To practice this part of the experiment, you lot will utilize a 100-mL round bottom flask as the distillation pot. You will need a number of receivers; it is best to apply test tubes. Measure into one examination tube about 4 mL of water. Use this sample to know how much liquid you need to obtain about iv mL of distillate during this part of the experiment. Continue collecting 4-mL samples until you have collected about 30 mL of distillate. Decide the refractive index of each collected sample, besides as determining the refractive alphabetize for pure ethanol and pure water.
- Obtain about l.0 mL of the l% (5/5) ethanol/h2o mixture, and pour into the distillation pot.
- Add together a few boiling stones.
- Plough on the heating mantel to obtain a steady boiling mixture.
- Monitor time and temperature during the entire distillation process
Beginning recording the temperature as soon every bit your sample begins to boil. Record the temperature every thirty sec. Collect your distillate into examination tubes. You should collect most four mL in each exam tube, but it is not necessary to mensurate each tube. As a comparison, add about iv mL of water into a examination tube. Collect almost the aforementioned amount of liquid into each of the tubes during the distillation procedure. Proceed recording the temperature until you stop collecting your samples. Collect most 30 mL of distillate.
You must plot your data past hand using graph paper, or y'all tin can utilise ChemWorks™ on the computers for inclusion in your lab notebook and written study. Y'all should have two plateaus, ane for the boiling point of the ethanol and the other for the humid signal of the water. Your graph for your lab written report and for your notebook must show these two plateaus.
Afterwards the distillation has finished, yous will accept a practiced determination for the boiling point of ethanol. Determine the refractive index for each of your samples.
Do not ever throw any glass chaplet away. At an expense of near 25 cents ($0.25) per glass bead, they are very expensive. Go along your glass beads in your fractionating cavalcade (add some tissue to the top to prevent spillage) until the side by side lab menses. Never throw abroad any drinking glass beads.
Part C: Fermentation and Distillation of Ethanol
Yeast ferment sugars to produce ethanol. Yous will utilise glucose (dextrose) as the sugar the yeast volition employ for booze production. Glucose has a tooth mass of 180 one thousand/mol. The tooth mass of ethanol is 46 grand/mol. The other product of fermentation is carbon dioxide, which we will non consider in the current experiment. The equation for this reaction is equally follows:
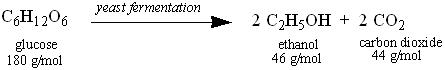
Based on the counterbalanced equation, ane (1) mole of glucose will yield two (2) moles of ethanol. Based on these tooth ratios, one mole of glucose is 180 g, and 2 moles of ethanol is 92 grams. On the other hand, one mole of sucrose will yield four moles of ethanol. Your Instructor volition inform y'all which carbohydrate you will be using.
- How many grams of alcohol would yous produce, if you started with 60.00 grams of glucose (MW=180.0)?
- If you lot started with sixty grams of sucrose (MW=342.three), how many grams of ethanol would you produce?
Based on the corporeality of sugar (glucose or sucrose) y'all actually started with (based on the amount y'all weighed out), what is the theoretical yield of alcohol that you should produce? Utilize this value to determine your percent yield subsequently you have finished this part of the experiment.
| Grams of glucose (or sucrose) fermented: _________________ g | Theoretical yield of ethanol: __________________ g |
Fermentation Setup
Set upward the fermentation container in the following mode:
- Place 60.00 g (0.33 mole) of glucose (or of sucrose) into a 250-mL Erlenmeyer flask (or the "beer-canteen fermented")
- Add together 175 mL of distilled h2o
- Add together 20 mL of the Pasteur's salts solution (see Footnote i for conception)
- Add two.00 g dry out yeast, that has been rubbed to a thin paste (to suspension upwardly yeast clumps) with about 10 mL of DI water
- Shake vigorously to mix the contents of the fermentation container (containing all reagents)
- Close the container with a safe stopper attached to a piece of glass tubing connected by a piece of latex tubing. The glass tubing at the end of the latex tubing volition be inserted into a tube containing water (2-3 mL DI water) and a minor amount of mineral oil (pour a niggling mineral oil to cover the water in the exam tube) to prevent evaporation.
Allow the fermentation mixture to stand at a temperature of 25-35oC until fermentation is consummate (about a calendar week is required).
Get-go Distillation of Fermentation Mixture
After the fermentation is complete, you lot volition distill the alcohol from the liquid mixture, including the solid, particulate cloth that is not soluble. Gently cascade all of the liquid into a 500-mL round-bottomed flask (checked out from the stockroom). The liquid will be somewhat cloudy, but this volition not interfere with the first distillation (do not ever let a distillation flask go dry, only this should exist of piddling concern here since we will only collect a fraction of the liquid via distillation).
Add a few (half-dozen-viii) humid stones to the flask. Attach a partial distillation column (filled with drinking glass chaplet, as described in Function B) to the top of the distillation flask, and attach the still caput and condenser unit. You lot exercise non need to monitor the temperature equally the outset distillate is mostly water, so the temperature will be about 100oC. Collect about l mL of distillate into a pre-weighed graduated cylinder (a fifty- or 100-mL graduated cylinder can be used).
After the first distillate has been collected, discover and record the collected volume, and counterbalance the graduated cylinder to decide the mass of collected distillate (total mass of cylinder containing the distillate minus the mass of the empty graduated cylinder). You should now calculate the density of the distillate, based on the mass of the distillate you but determined divided past the volume collected. From the Table of Densities of aqueous ethanol solutions, given in Footnote ii, calculate the mass of alcohol collected in this first distillate.
To make your determination of the amount of ethanol isolated, you can estimate your percent alcohol using the table at the end of this experiment. This table shows you several ways to determin the corporeality of ethanol in your sample based on density. The easiest column to use is the first column which shows the mass pct of alcohol (% by mass) as follows:
Using the density of your distillate from the above calculations, determine the per centum alcohol ( you may have to extrapolate (east.1000., Calculate the percent yield of ethanol produced in the fermentation, based on the theoretical amount of ethanol that would be produced from the starting amount of glucose (each glucose molecule produces two ethanol molecules). This "crude" distillate volition be used in a second fractional distillation.
An additional table is bachelor, using an interpolative procedure to determin percentages which do not fall inside the larger percent range shown in the table below (percentages are in 5% increments). The new tabular array (which is an Adobe pdf document) shows you how to get to the per centum values, based on density, in unit of measurement values, e.thou., 45%, or 46%, or 47%, etc. The highlighted values in this pdf document shown how to determine percent concentration values between 45% and fifty% ethanol. You can download and print this document at this URL of a pdf certificate.
Discard the balance left in the distillation pot, which contains by and large water.
I tin can obtain 95% ethanol (but not 100% ethanol; do yous know why? What is an azeotrope?) from the dilute alcohol mixture obtained during the first distillation. For the second distillation, monitor the boiling point carefully, as you should collect the cloth that distills at a temperature of 78-82oC; if too petty distillate is obtained in this range, continue the distillation and collect the fraction boiling at 82-88oC.
Prior to doing your 2d distillation, empty the glass beads from the fractioning column into a beaker y'all will wash them yourself with lather and water, and then reuse them. Also, you need to wash with soap and water each of the following pieces of glassware:
- Fractionating column
- Still head
- Condenser column
You tin now re-assemble your distillation setup for a fractional distillation, and and so follow the procedure for collecting and analyzing your 2nd-distillate liquid. At the stop of the experiment, put the used glass chaplet into a chalice on the Instructor'south demote to be done by the Stockroom Personnel. Do not throw the drinking glass bead away.
Procedure for drove of ethanol via a second distillation:
- Add the alcohol-water mixture from the start distillation (afterwards weighing and determining its density) to a 100-mL round bottom flask.
- Add a few boiling stones to your circular-bottom flask (distillation pot) to maintain a slow, steady humid.
- Commencement monitoring the temperature equally shortly as yous plough on the estrus, or at least prior to the solution boils. Monitor the temperature at regular intervals, usually every minute, until you end collecting your samples.
- Beginning to collect iv-mL samples (using y'all conical vial in the organic kits or a test tube) until the temperature rises significantly above the normal 78-82oC temperature, every bit described above. It is notwithstanding permissible to collect above the 82oC degree range, but your samples incorporate more and more h2o. Notwithstanding, if you take not collected at least iv-5 samples, each of about 4-5 mL volumes, continue to collect samples (regardless of temperature) until at least 20 mL of solution is collected. Based on theoretical yeields, and bodily lab experience, you could have produced 30-36 mL of ethanol during your fermentation, so collectin at least xx mL is non out of reason.
- Exercise non determine a refractive, simply y'all must make up one's mind a density for each sample. To exercise this, collect about 4-5 mL in a conical vial (as mentioned in a higher place), and then immediate transfer the contents of the vial into a pre-weighed 10-mL graduated cylinder. After transfer of collected fabric into the 10-mL graduated cylinder, reattach the vial to the vacuum adapter, and collect another 4-five mL sample.
- Decide the mass and volume of the sample in the 10-mL cylinder to summate a density of the sample. After determining the mass and book, transfer its contents into a pre-weighed 50-mL graduate cylinder to collect all samples for an overall density and yield of ethanol for the unabridged experiment.
- After you have analyzed individually the iv-mL samples, combine your 4-5-mL collected distillate samples into the 50-mL graduated cylinder, the full volume, the mass of the sample in the l-mL cylinder, yous can determine the density.
- From the density but determined (footstep #7), determine the full mass of ethanol nerveless during the 2nd fractional distillation. No need to determine a refractive index equally density (and total mass of solution) volition be our criterian for yield.
- Determine percentage yield based on the overall yield from step #8, by dividing the actual yield by the theoretical yield times 100 to get a percent yield.
Based on the amount of collected alcohol, its density, what is the percent yield? What is the theoretical yield?. Your instructor will help you determine the percentage of alcohol based on your refractive alphabetize and density determinations.
Do non throw whatever glass chaplet away. At an expense of nearly 25 cents ($0.25) per glass bead, they are very expensive. After all distillations are completed, pour your drinking glass chaplet into a big beaker on your instructors demote, then they can exist cleaned. Never throw glass chaplet abroad.
| Compound | MW | Amount | mmol | mp | bp | Density | η D | msds |
|---|---|---|---|---|---|---|---|---|
| 2-propanol (isopropyl alcohol) | 60.ane | 50 mL | -88.five | 82.5 | 0.78505 | i.3772 | msds | |
| Dextrose (glucose) | 180.16 | 50.0 g | 277.53 | 146 | --- | i.54 | msds | |
| Sucrose | 342.3 | l.0 one thousand | 146.07 | 160-186 | --- | ane.59 | msds | |
| Ethanol | 46.0414 | -114.1 | 78.5 | 0.viii | 1.3614 | msds | ||
| Compound | thou/mol | grams or mL | 10-3 1000 | oC | oC | g/mL | ηD | msds |
Source: https://home.miracosta.edu/dlr/210exp5.htm
0 Response to "How Do You Know if Distillation Is Successful"
Post a Comment